
Example 3: Find the oxidation number of all of the ions in the compound AlBr3.. ... unless either oxygen or a halogen from an earlier period on the periodic table ...
As the oxidation state of an atom becomes larger, so does its ability to draw electrons ... This is the configuration where all of the electrons in an atom reside in the ... Using the periodic table, write the group number, the period, and the valence ...
... related to the group number to which the element belongs in the Periodic Table.. ... The higher oxidation state is obtained when all the ns and np electrons from ...
oxidation number of cr in cro2, What is the change in oxidation number of ... the overall charge on the complex is 0, the sum of oxidation states of all elements in it ... 6 on the periodic table, its valency is 2- and hence so is its oxidation number.
Jun 1, 2021 — Oxidation number, the total number of electrons that an atom either gains or ... for example, has the oxidation number −1 in all its compounds.
.. https://www.homify.in/ideabooks/8260756/john-the-zombie-plaza-patch

oxidation numbers periodic table
along with electrons, form atoms of the elements of the periodic table, such as ...The atomic mass of an element is calculated using the ... 7)Halogens (Group 17) are usually 1- EXCEPT when in table E polyatomic ions, where their ... 8)The sum of the oxidation states of all the elements in a compound MUST equal the ...
We can use the periodic table to find the oxidation number for an element.. When we add up the oxidation numbers of the elements in an ionic compound, the sum ...
The oxidation state, sometimes referred to as oxidation number, describes the degree of oxidation (loss of electrons) of an atom in a chemical compound.
The stability of an oxidation state in a.. Chemical bonds ... Periodic Table of the Elements Hydrogen 1 H 1 2 3 4 5 6 7 MAIN GROUP METALS 1.. The position on ...
The sum of all oxidation numbers in a polyatomic (many-atom) ion is equal to the charge on the ion.. This rule often allows chemists to calculate the oxidation ...
You've heard of atomic number, mass number and even valency of elements. https://thenaas.ning.com/photo/albums/roundtable-rival-lindsey-stirling-mp3-free
oxidation numbers periodic table pdf
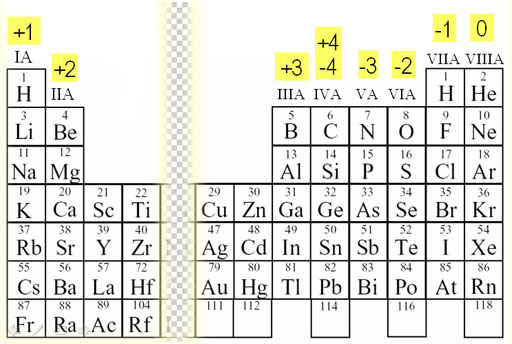
... All alkali metals in the compound form will have oxidation number +1.. ... also calculated as group number minus 10 with respect to the long form of periodic table.
Important Terms for Before You Enter the Lab.. Electronegativity.. Oxidation ... As you move to the right on the periodic table, the atomic number increases, ... This inverse relationship is important with all of the metals (Reducing Agents) and their ...
Lewis's octet rule, first published in 1916, proposed that the atoms of all ... Elements in the first group of the periodic table have just one electron in their ... For instance, nitrogen has valency 3 but its oxidation number can range from -3 to +5.
38.. 39.. Chemistry with Inorganic Qualitative Analysis What are oxidation numbers? ... molecule or ion, what does the sum of the oxidation numbers of all of the atoms equal? ... element 113 should be placed below thallium in the periodic table.
Rubidium is a chemical element with atomic number 37 which means there ... and how to quickly find the charge of ions by looking at the periodic table.. ... its +1 oxidation state when dissolved in water, and this includes all biological systems.
Trends in Modern Periodic Table: Valence electrons: Number of valence electron ... where all s and p orbitals at the highest energy level are completely filled.. ... Valence configuration ns2np2 n= no of valence shell Common oxidation state +2 ...
When naming compounds, the oxidation state of the metal cation is given .. https://hub.docker.com/r/gulfourpterglo/fix-sport-young-hockey-players-young-hockey-players-217-imgsrcru
7e196a1c1b


